Important information and recommendations regarding the European Medical Device Regulation (MDR)

Implementing the MDR in accordance with VITA quality standards
1. VITA veneering and CAD/CAM materials
All VITA veneering and CAD/CAM materials bear a UDI-compliant Health International Bar Code (HIBC) that contains all the required information, either in digital form or in plain text. All units produced (e.g., bottles or blocks) are labeled with a batch number. The scannable barcode is provided on the primary packaging of the product.
2. VITA prosthetic teeth
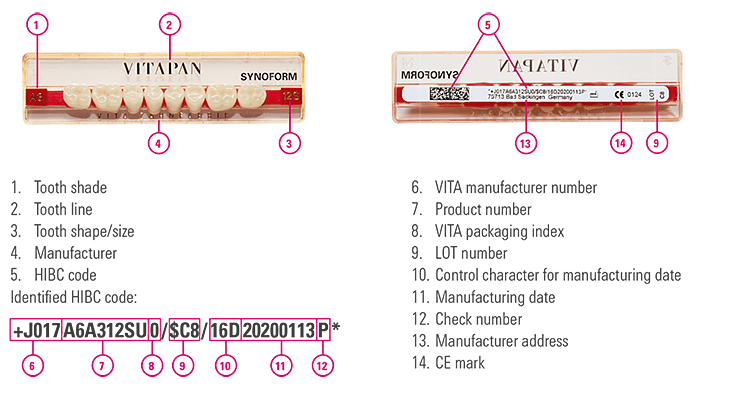
Our tooth sets are also labeled with a UDI-compliant Health International Barcode. Every tooth set includes a verified and approved tooth batch that ensures correct batch traceability. As the risk class is low, batch identification is not required for each individual tooth.
Tooth set description VITAPAN EXCELL, VITA LINGOFORM, VITAPAN PLUS, VITAPAN, VITAPAN CUSPIFORM, VITA PHYSIODENS

Tooth set description VITAPAN SYNOFORM, LUMIN VACUUM, VITA PHYSIODENS
VITA tips and recommendations
Documentation options
To facilitate documentation, we recommend using a documentation software. Using a barcode reader, all UDI-compliant HIBC information on the medical devices can be scanned, transferred to the documentation soft-ware and saved.
Manual documentation of the product number and batch to ensure sufficient traceability, can also be carried out without a barcode reader.
We do not attach adhesive labels, as this is not a sustainable solution and would require management of vast quantities of labels, given the large number of components.
Declarations of conformity
You can conveniently download and manage declarations of conformity in our MyVITA online portal.
Simply register at www.vita-zahnfabrik.com/MyVITA-Register
In the "Declarations of conformity" section, you can access the service "Automatic updates for your declarations of conformity." If requested, your selected VITA declarations of conformity are updated automatically. You will also be informed via email when a new version is available.
